- 1/5
- 2/5
- 3/5
- 4/5
- 5/5
- QC
- Environmental Monitoring
- Plates
- AAV6 Plasmid
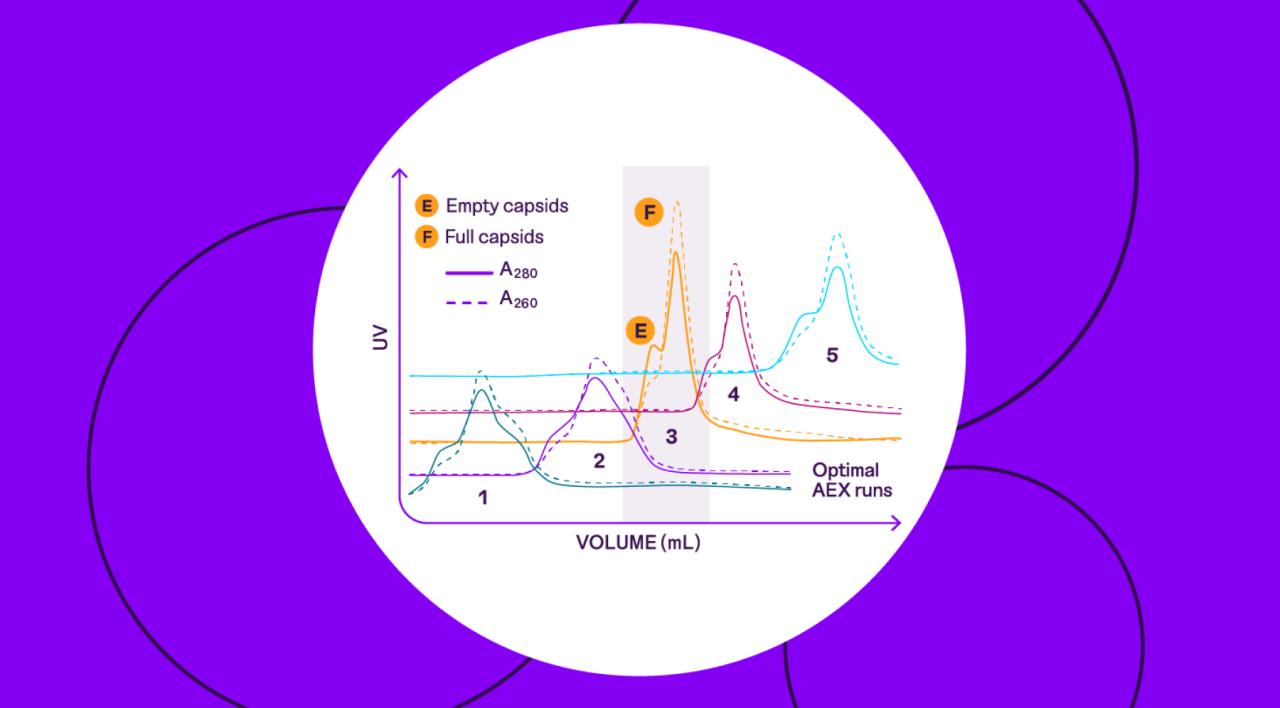
- AEX
- Culture
- Extraction and Purification
- Analytics
A trusted partner that
can help you scale
With over 25 years experience manufacturing custom reagents, we have finnessed the rapid turnaround of customer-specified products. Our new state-of-the-art facility has increased our footprint to over 200,000 ft2, enabling us to better serve our customers by providing the scale and flexibility needed for GMP manufacturing.
Find out what’s possible
Talk to our consultants today to find the ideal off-the-shelf or custom solution that can help you achieve your goals.
Our team is here to help
Get in touch:
1.800.209.4488
Monday to Friday
8:00AM–5:00PM PT